Protocols and mandates cannot govern all of medical practice.
Few better examples of cookbook failure exist than the decision-making surrounding the Medtronic Sprint Fidelis lead.
You may know the story already. Here’s my brief summary:
In the early 2000’s, Medtronic released a family of defibrillator leads that were promoted as thinner and easier to maneuver. They even made them pretty to look at. And they delivered on their promise; they handled beautifully, which made the implant surgery easier. I used them; so did many other docs, as more than 250,000 were implanted.
The problem was the leads weren’t durable; they failed way too frequently. Many of these failures were unexpected and sudden. Some patients died, many more suffered painful shocks. Soon after their widespread acceptance, though some would argue not soon enough, Medtronic pulled them from the Market in 2007.
Since the majority of these leads were placed in 2005-2007, and ICD generators last about 5-6 years, many affected patients are now coming up for elective ICD generator change. This brings to the fore some interesting problems.
Should we place a new lead at the time of elective surgery? The knee-jerk answer is yes. But placing a new lead increases the risk of surgery.
What if the lead hasn’t shown any problems?
What if the patient is pacing-dependent? In this case, abrupt lead failure could be catastrophic, but so would a surgical complication.
Today, a report in Circulation provided some important data on this vexing dilemma. The trial followed 1023 patients with Sprint Fidelis leads from 2001-2009. The researchers from three-centers (one in Boston and two in Minnesota) reported six relevant findings:
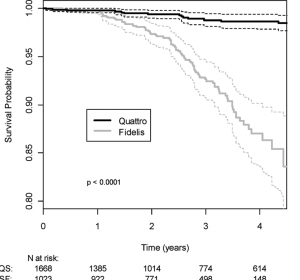
- The failure rate for Sprint Fidelis leads was 2.81% per year–versus 0.43% for the currently-used Medtronic Quattro ICD lead.
- Four-year lead survival was 87%–versus 98.7% for the Quattro lead. (Translation: At minimum, greater than one in ten patients can expect their Sprint Fidelis lead to fail.)
- 42% of lead failures resulted in inappropriate shocks.
- The lead failure rate doesn’t plateau over time. (See Figure)Â The longer these leads remain, the lower their survival.
- Young patients, female patients, those with hypertrophic cardiomyopathy (abnormally thick heart), and other unusual electrical diseases of the heart were at higher risk of lead fracture. The young are thought to be at higher risk of failure because their more vigorous youthful lives put more and tear on the vulnerable lead insulation.
- This study reported higher failure rates than those previously released by Medtronic.
Real-life patients with Sprint Fidelis leads illustrate the darned if you do and darned if you don’t doctrine. One doctor may see the risk of lead failure as too high, while another sees the risk of surgery as worse. Some patients become disabled after shocks, others pay them little mind. Some operators are highly skilled at placing new leads, for others it’s a more serious challenge.
This current study helps confirm my bias for replacing the Sprint Fidelis lead at the time of elective surgery, especially in a younger patient with a normally contracting heart muscle.
But we are still a long way off from applying the cookbook principle to this problem.
JMM